|
7/9/2023 0 Comments Platinum notes 4 download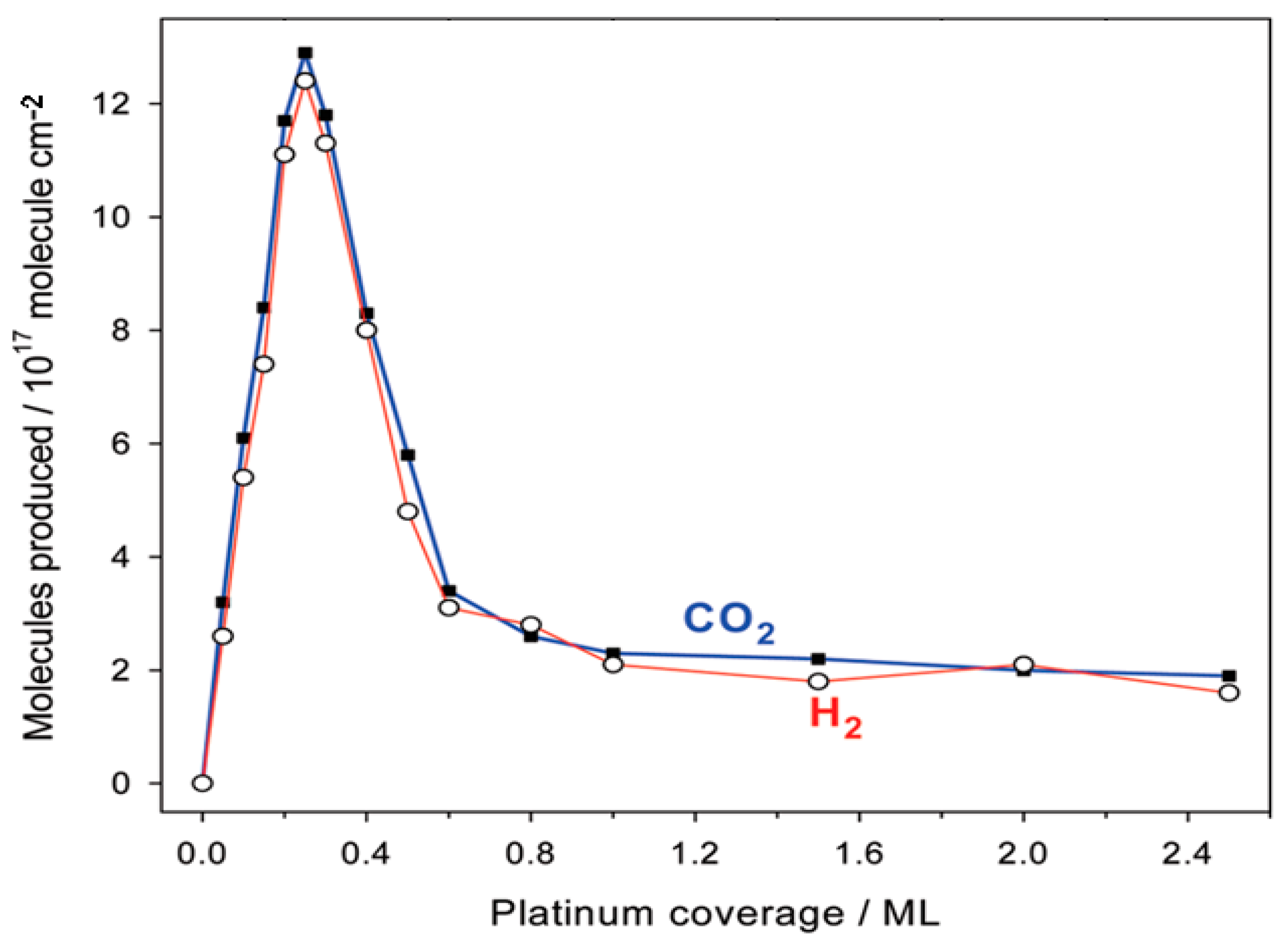
The blue colour of the solution (Cu 2+ ) fades then turns colourless A red brown solid, copper metal is deposited. The blue colour of the solution (Cu 2+) fades then changes to green (Fe 2+) The procedure is repeated with 1M magnesium sulphate solution instead of CuSO 4(aq).The procedure is repeated with fresh samples of CuSO 4 with Zn, Mg, and Cu powders. Any observations and temperature change are determined and recorded. In the solution, a spatula end-full of iron fillings is added. 5 cm 3 of 1M CuSO 4(aq) is put in a test-tube and its temperature recorded.Electrochemical reactions involve transfer of electrons and are essentially REDOX reactions.ĭisplacement and REDOX Reactions Experiment 1:- Displacement Reactions among Metals.Electrochemistry is the chemistry of electrochemical reactions which deal with the relationship between electrical energy and chemical reactions.Factors Affecting Electrolysis and Electrolytic Products.Terms Used in Describing Oxidation-reduction.  
0 Comments
Leave a Reply. |


 RSS Feed
RSS Feed
